�
What is Age-Related Macular Degeneration?
|
Age-Related macular degeneration (AMD) is a leading cause of blindness and vision loss in the United States. In 2019, it was estimated that 18.34 million Americans 40 years and older were living with early-stage AMD, and 1.49 million with late-stage AMD. AMD is the leading cause of blindness in people living in developed countries, typically affecting approximately 28% of individuals 75-85 years of age. As the “baby boomer” generation grows older, it is expected that the number of people living with AMD will rise significantly to 288 million people by 2040 worldwide [1]. AMD is a disease of the retina, more specifically the macula. The macula is a component of the retina which facilitates the processing of visual stimuli located directly in front of ones field of view, also referred to as central vision (figure 1). Although complete blindness is not associated with AMD, loss of central vision can make reading, driving, or even recognizing family members difficult. AMD is a progressive disease, meaning symptoms typically worsen over time; however, the rate of progression varies from individual to individual [2].
|
Figure 1: The Amsler grid is a typical diagnostic tool used to detect problems with central vision. A normal eye would see the Amsler grid as shown on the left. Someone with AMD, or another retinal disease affecting central vision, would see the Amsler grid as shown on the right.
|
It is believed that AMD results from the inability to regulate the intensive metabolic processes of retinal cells, leading to the accumulation of by-product waste. In normal cells, this by-product waste can be broken down by the body. In patients with AMD, by-product waste accumulates as we age, and mitochondria become less effective at "cleaning up" this waste. This accumulation causes small deposits called "drusen" to form, which can be defined as accumulated proteins and waste products within the retina. Drusen deposits interfere with the retina's ability to supply its cells with oxygen and nutrients, thus causing cell death and central vision loss [3]. There are two forms of AMD: dry and wet. Dry AMD is the most common form of AMD (about 90% of all cases), and is characterized by drusen (figure 2). Importantly, cases of AMD always start out in the dry form; however, can progress to a much more severe case known as wet AMD. Wet AMD occurs through angiogenesis, or the overgrowth of blood vessels (figure 3). In severe instances, these blood vessels can burst, causing extreme damage to retinal integrity.
Dry AMDFigure 2: Drusen deposits in the back of the retina, associated with dry AMD
Cases of AMD always start off as dry. Dry AMD occurs due to drusen deposits, and is associated with cell death and retinal thinning. Dry AMD progresses slowly with age, accompanying a gradual decrease in central vision. There is currently no treatment available for dry AMD. |
Wet AMDFigure 3: Angiogenesis, characteristic of wet AMD, causes structural damage to the macula.
In an attempt to compensate for a lack of nutrients and oxygen, due to drusen deposits, the body may form blood vessels in a process called angiogenesis. When these blood vessels form, they can grow into the retina and cause structural damage. Along with this, blood vessels can burst and leak blood into the retina, causing direct structural damage. Wet AMD causes a significantly faster rate of vision loss. Despite this, treatments are available to slow down angiogenesis, however, a dry AMD phenotype is impossible to reverse. |
|
Figure 4: Progression of AMD vision loss at different stages. From left to right: Early stage (at risk/early signs), intermediate, and late-stage (advanced).
|
Stages of Age-Related Macular Degeneration: AMD consists of three stages: early-stage, intermediate, and late-stage (Figure 4).
|
|
So What Causes AMD? The etiology of AMD is largely unknown, especially with regard to genetic predisposition. AMD has, however, been associated with numerous risk factors, many of which are linked to oxidative stress. Oxidative stress is characteristic of cell degradation via the damaging effects of free oxygen radicals (Figure 5), and is a large contributor to genome instability as well as protein degradation. It is generally believed that, during the aging process, damaged mitochondria produce less ATP and cause ROS levels to accumulate. This accumulation of ROS leads to exaggerated amounts of oxidative stress, thereby inducing an energetic crisis within the cell that triggers neurodegenerative disorders and accelerates the aging process [5]. In the context of photoreceptor health, for example, advanced glycation end-products (AGEs) and carboxyethylpyrrole adducts are oxidative modifications of the photoreceptor fatty acid tips, and have both been identified within isolated drusen samples of patients with AMD [6]. Because oxidative protein modifications have been identified within the proteins of drusin deposits, the hypothesis that oxidative stress plays a key role in the age-related prognosis of AMD is becoming increasingly compelling [7]. Most of this increase in oxidative stress is due to the normal aging process, however, other risk factors such as poor diet, lack of exercise, and genetics play a role in the metabolic activities of the retina, and can increase the risk of developing AMD. |
Figure 5: The effect of oxidative stress, or reactive oxygen species, on the integrity of a cell. A similar pattern can be illustrated by the oxidation of a tomato.
|
Prevention and Treatment
Natural PreventionThe first line of defense against AMD is the body's natural defense. When ROS accumulate in the back of the retina, certain proteins can sequester these oxygen radicals and prevent damage to the retina. This is known as the "antioxidant defense response". Within this process, the cell detects a high abundance of ROS and induces the production of antioxidant defense proteins. Similarly, antioxidants can be consumed as a micronutrient in our diet (figure 6). Although antioxidant micronutrients (from food) and antioxidant proteins (synthesized intracellularly) serve similar functions, they are derived from different origins. One such antioxidant protein is PGC-1α. PGC-1α is capable of inducing downstream antioxidant protein expression so that ROS are sufficiently sequestered, thereby minimizing damage to the retina. Over time, however, our ability to regulate this process becomes less efficient. ROS can accumulate as we age and therefore increase the risk of developing AMD at a later age in life. When our body is no longer able to naturally regulate these processes, we must look to potential therapeutic options to reverse or prevent the death of photoreceptor cells.
|
Figure 6: Foods that are high in antioxidants. AMD has been associated with nutritionally starved diets, and eating foods rich in antioxidant micronutrients could help prevent blindness later in life [8].
|
|
Figure 7: A model showing the in vitro creation of functional RPE cells from pluripotent stem cells. These stem cells are then transplanted into an AMD-affected retina to slow down or reverse photoreceptor death.
Figure created using BioRender. |
Stem Cell TherapyIn 2019 and 2020, there was a major leap forward when the first therapeutic intervention for AMD was successfully implemented in a patient. Currently, the only recognized treatment for AMD uses stem cell transplantation to prevent the death of photoreceptors. In this process, retinal pigment epithelium (RPE) cells are derived from pluripotent stem cells in vitro. These cells are then transplanted into the retina where they can reverse or prevent the death of neighboring photoreceptors (Figure 7). AMD degrades the RPE layer of the retina first, subsequently leading to the death of photoreceptors. Therefore, by replenishing the RPE layer with stem cells, the death of photoreceptors can be slowed, halted, or even reversed. The effectiveness of this treatment is dependent on the quality, survival, and purity of the stem cells, along with the body's immune environment [9].
|
Future Therapeutic Direction
|
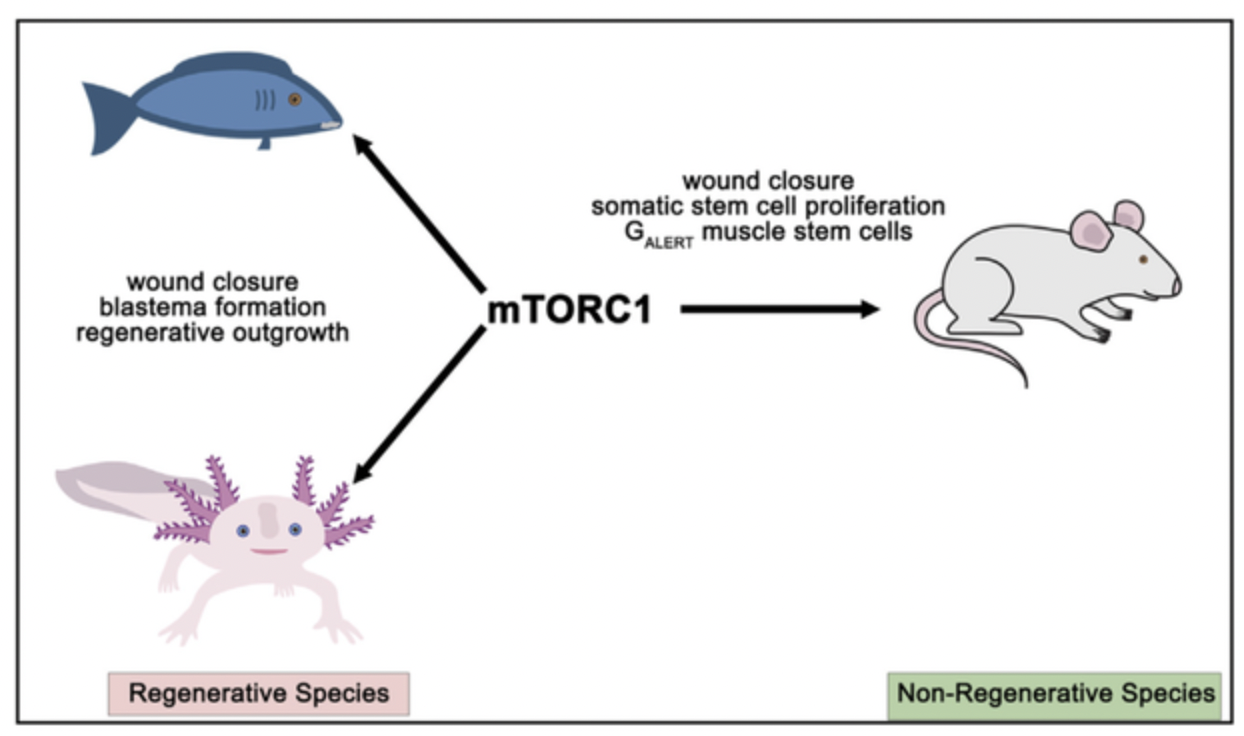
Our body's natural prevention of AMD via the antioxidant defense response and stem-cell based therapies both yield great potential in reducing, or reversing, vision loss. Each mechanism, however, has its own downsides. We are unable to manually control the body's natural antioxidant defense response (outside of our diet), and stem-cell based therapies are limited by surgical techniques and stem cell quality. One organism has found a way to combine these two mechanisms: the zebrafish (Danio rerio). In zebrafish, metabolic regulatory pathways can regenerate individual cells, tissues, and even entire organs (figure 8). These pathways are modulated via mTOR, in parallel with other proteins, such as PGC-1α and NR5A2 [10]. This mechanism will be the main focus of this study, with the goal of identifying regenerative mechanisms in zebrafish that could be used to develop therapeutic interventions in humans with AMD. The gene PGC-1α, and its respective protein, is a key player in the activation of antioxidant defense proteins and regeneration-signaling proteins in zebrafish. It is unknown, however, how PGC-1α-regulated metabolism induces photoreceptor regeneration in zebrafish.
To learn more about why zebrafish are the best model organism for this study, visit the page "Model Organism" |
Figure 8: Zebrafish, like axolotl, are able to regenerate entire body parts and tissues much more effectively than non-regenerative organisms like mice and humans. This is due to metabolic regulatory cascades intertwined with the proteins PGC-1α, mTORC1, and NR5A2.
|
Public Resources for AMD
National Institute of HealthNational Eye Institute
|
Center for Disease ControlNational Prevalence of AMD
|
References
1. Ferrington, Deborah A et al. “Altered bioenergetics and enhanced resistance to oxidative stress in human retinal pigment epithelial cells from donors with age-related macular degeneration.” Redox biology vol. 13 (2017): 255-265. doi:10.1016/j.redox.2017.05.015
2. Rein DB, Wittenborn JS, Burke-Conte Z, et al. Prevalence of Age-Related Macular Degeneration in the US in 2019. JAMA Ophthalmol. 2022;140(12):1202–1208. doi:10.1001/jamaophthalmol.2022.4401
3. “Age-Related Macular Degeneration (AMD).” National Eye Institute, U.S. Department of Health and Human Services, 22 June 2021, https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/age-related-macular-degeneration.
4. InformedHealth.org [Internet]. Cologne, Germany: Institute for Quality and Efficiency in Health Care (IQWiG); 2006-. Age-related macular degeneration (AMD): Overview. 2006 May 22 [Updated 2018 May 3]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK315804/
5. Hanna, Joseph, Luke Ajay David, Yacine Touahri, Taylor Fleming, Robert A. Screaton, and Carol Schuurmans. “Beyond Genetics: The Role of Metabolism in Photoreceptor Survival, Development and Repair.” Frontiers in Cell and Developmental Biology 10 (2022). https://doi.org/10.3389/fcell.2022.887764.
6. Crabb, John W et al. “Drusen proteome analysis: an approach to the etiology of age-related macular degeneration.” Proceedings of the National Academy of Sciences of the United States of America vol. 99,23 (2002): 14682-7. doi:10.1073/pnas.222551899
7. St-Pierre, Julie et al. “Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators.” Cell vol. 127,2 (2006): 397-408. doi:10.1016/j.cell.2006.09.024
8. Montgomery MP, Kamel F, Pericak-Vance MA, Haines JL, Postel EA, Agarwal A, Richards M, Scott WK, Schmidt S. Overall diet quality and age-related macular degeneration. Ophthalmic Epidemiol. 2010 Jan-Feb;17(1):58-65. doi: 10.3109/09286580903450353
9. O'Neill, Helen C et al. “Advancing a Stem Cell Therapy for Age-Related Macular Degeneration.” Current stem cell research & therapy vol. 15,2 (2020): 89-97. doi:10.2174/1574888X15666191218094020
10. Bellezza, Ilaria. “Oxidative Stress in Age-Related Macular Degeneration: Nrf2 as Therapeutic Target.” Frontiers in Pharmacology 9 (2018). https://doi.org/10.3389/fphar.2018.01280.
2. Rein DB, Wittenborn JS, Burke-Conte Z, et al. Prevalence of Age-Related Macular Degeneration in the US in 2019. JAMA Ophthalmol. 2022;140(12):1202–1208. doi:10.1001/jamaophthalmol.2022.4401
3. “Age-Related Macular Degeneration (AMD).” National Eye Institute, U.S. Department of Health and Human Services, 22 June 2021, https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/age-related-macular-degeneration.
4. InformedHealth.org [Internet]. Cologne, Germany: Institute for Quality and Efficiency in Health Care (IQWiG); 2006-. Age-related macular degeneration (AMD): Overview. 2006 May 22 [Updated 2018 May 3]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK315804/
5. Hanna, Joseph, Luke Ajay David, Yacine Touahri, Taylor Fleming, Robert A. Screaton, and Carol Schuurmans. “Beyond Genetics: The Role of Metabolism in Photoreceptor Survival, Development and Repair.” Frontiers in Cell and Developmental Biology 10 (2022). https://doi.org/10.3389/fcell.2022.887764.
6. Crabb, John W et al. “Drusen proteome analysis: an approach to the etiology of age-related macular degeneration.” Proceedings of the National Academy of Sciences of the United States of America vol. 99,23 (2002): 14682-7. doi:10.1073/pnas.222551899
7. St-Pierre, Julie et al. “Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators.” Cell vol. 127,2 (2006): 397-408. doi:10.1016/j.cell.2006.09.024
8. Montgomery MP, Kamel F, Pericak-Vance MA, Haines JL, Postel EA, Agarwal A, Richards M, Scott WK, Schmidt S. Overall diet quality and age-related macular degeneration. Ophthalmic Epidemiol. 2010 Jan-Feb;17(1):58-65. doi: 10.3109/09286580903450353
9. O'Neill, Helen C et al. “Advancing a Stem Cell Therapy for Age-Related Macular Degeneration.” Current stem cell research & therapy vol. 15,2 (2020): 89-97. doi:10.2174/1574888X15666191218094020
10. Bellezza, Ilaria. “Oxidative Stress in Age-Related Macular Degeneration: Nrf2 as Therapeutic Target.” Frontiers in Pharmacology 9 (2018). https://doi.org/10.3389/fphar.2018.01280.
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison
Contact:
Paul Lyons
[email protected]
Last Edited on January 24th 2023
www.genetics564.weebly.com
Contact:
Paul Lyons
[email protected]
Last Edited on January 24th 2023
www.genetics564.weebly.com